Recently, Prof. Zhengcao Li's group at School of Materials Science and Engineering of Tsinghua University, and Marnix Wagemaker from Department of Radiation Science and Technology at Delft University of Technology published a research article entitled " Operando monitoring the Lithium spatial distribution of Lithium metal anodes" in Nature Communications. In this article, Neutron Depth Profiling (NDP) was combined with isotope method to quantitatively analyze the spatial heterogeneity of Li in the plating and stripping process of lithium metal cells, which paves the way to the development of safe and reversible Li metal anodes.

Figure 1. Principle of operando neutron depth profiling of Li-metal plating and stripping. a principle and schematic setup of operando neutron depth profiling (NDP). b Operando NDP measurements of four plating and stripping cycles at 1.0 mA/cm2 current density.
Li metal has the highest specific capacity (3860 mAh/g), very low density (0.59 g/cm3), and the lowest redox potential (−3.04 V vs. standard hydrogen). For these reasons, Li-metal is recognized as the ultimate anode. Moreover, the cathode alternatives that are intensively studied include the Li-S and Li-O battery systems, and their theoretical specific capacity is much higher than existing commercial lithium-ion battery systems, which will become the most promising high-density energy storage system. However, the major challenges for Li metal anodes are safety and cycleability. Firstly, the safety risks associated with dendrite formation is directly related to its tendency to be deposited in a dendritic and mossy form. Then, when dendrites penetrate the separator/electrolyte and reach the cathode, this causes an internal short-circuit that may induce rapid spontaneous discharge and consequential safety hazards. In addition, metallic lithium is highly reactive and easily reacts with the electrolyte to form an inactive solid electrolyte interface (SEI) and cause capacity decay. Therefore, how to effectively inhibit the growth of lithium dendrites is the key to realize the practical application of lithium metal anodes.
Aiming at preventing, suppressing, or blocking dendrite formation is mainly based on our current understanding of electrochemical Li-metal plating, the evolution of the complex SEI/Li-metal microstructure should be considered to assess the safety risks and energy storage efficiency of Li-metal batteries. Developing understanding of the Li anode microstructures is challenged by the difficulty to detect Li, both its quantitative distribution and chemical form, in particular during realistic battery operation. Here we apply NDP with isotope 6Li, complementary to microscopic and spectroscopic techniques, providing the spatial distribution/density of lithium during plating and stripping. The evolution of the Li-metal density-profile is shown to depend on the current density, electrolyte composition and cycling history, and allows monitoring the amount and distribution of inactive lithium over cycling. The morphology of the SEI strongly depends on the initial current rate, which influences the Li-metal morphology as well as the reversible Coulombic and Li efficiency on subsequent cycles. Unexpectedly, insertion and extraction of a small amount of reversible Li is observed in the copper current collector. It is confirmed that the copper grain boundary plays a dominant role by in-situ XRD analysis and molecular dynamics simulation based on first principles.
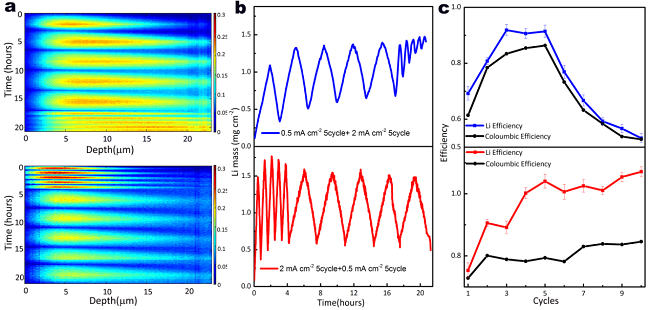
Figure 2. Evolution of the total amount of Li. a Operando NDP for five plating/stripping cycles. b Integrated amount of Li from the operando NDP experiments in a. c Coulombic efficiency and Li efficiency during the cycling.